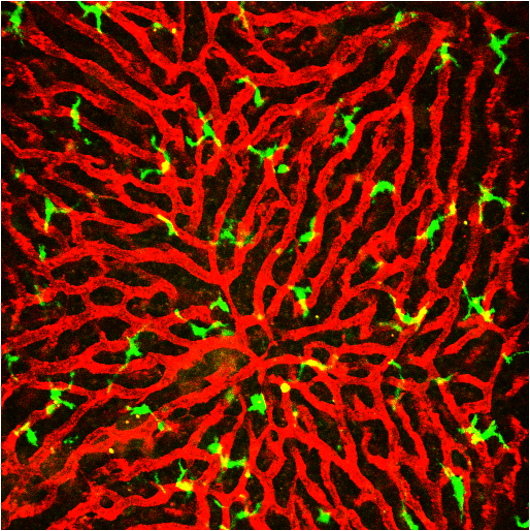
Intravital longitudinal imaging of hepatic lipid droplet accumulation in a murine model for nonalcoholic fatty liver disease
System Used:
Intravital Microscopy
Introduction:
In recent years, there has been a rapidly increasing interest in nonalcoholic fatty liver disease (NAFLD) with a high prevalence in the world. NAFLD has been attributed to central obesity, type 2 diabetes, and insulin resistance, which suggests NAFLD is a hepatic manifestation of metabolic syndrome. The most common form of NAFLD is hepatic steatosis, accumulation of excess fat in liver, which is the first histologic feature of the early stage of NAFLD. To more efficiently investigate the unknown cellular and molecular mechanisms in the pathogenesis of NAFLD in a complex in vivo hepatic microenvironment, a repetitive and longitudinal intravital visualization of the liver during NAFLD progression from the hepatic steatosis stage is a highly desirable approach. In this work, using a custom-built intravital confocal microscopy system [25–30], we successfully visualized individual LDs in hepatocytes in the liver of live mice in vivo with intravenous injection of SF44.
(Published info. : Biomedical Optics Express Vol. 11, Issue 9, pp. 5132-5146 (2020))
Instrument :
IVM-C (Intravital Confocal Microscopy system)
Methods:
To induce nonalcoholic steatosis and steatohepatitis, mice were fed with a methionine and cholinedeficient (MCD) diet (Envigo, USA). To observe temporal LD dynamics during the transition from normal liver to nonalcoholic hepatic steatosis, 4 groups of mice fed MCD with different period; 2, 7, 14, 21 days. The mice as control were fed with normal diet for the same period. 3 mice were used for each experiment group. Small incision with size of 10 mm was made on skin and peritoneum, then the left lobe of liver was exteriorized and positioned on a wet gauze to reduce motion artifact by separating other tissues near left lobe. The cover glass attached with a customized heater and commercial thermometer was placed on the exteriorized left lobe of liver, which maintain the temperature of local liver tissue constant at 36 °C during the intravital imaging. In addition, using a commercial homeothermic heating pad (RightTemp Jr., Kent Scientific) for small animal, the body temperature of the anesthetized mouse was also maintained at 36 °C during the intravital imaging. To prevent the surface of the exteriorized liver tissue from being dry, warmed saline was continuously supplied during the imaging.
Results & Discussion:
To perform the quantitative analysis of the dynamic changes in hepatic LD development during the progression of NAFLD, volumetric 3D rendered images were generated using in vivo z-stack images longitudinally acquired at 2, 7, 14 and 21 days of the MCD diet. As the MCD induction period (2, 7, 14 and 21 days) increased, we observed that the number of individual LDs in hepatocytes decreased, while the size of LDs gradually increased. The IVM system is optimized system for ultrafast cellular / molecular imaging (~100fps) with sub-um resolution in various organs and tissues of living small animal models. The IVIM Technology’s fully integrated all-in-one system can be used for long-term and repetitive in vivo imaging of living animals.