White Paper Highlight: An Efficient and Reliable Method to Measure SpO₂ in Rodents
Importance
Peripheral oxygen saturation (SpO₂) assesses blood oxygenation and directly measures efficiency of oxygen delivery to vital organs1. Accordingly, low SpO₂ is independently associated with increased mortality primarily driven by diseases like heart failure, COPD, asthma, pulmonary embolism and hypertensions2,3. Even if not directly researching these pathologies, SpO₂ levels are vital to monitor, especially since low SpO₂ can cause off-target effects to other systems, like the brain and the heart due to their high-metabolic demands4.
Using pulse oximeters are a reliable and non-invasive method of quickly monitoring this vital5. A normal, healthy SpO₂ reading is within 95% – 100%. Any level below 95% is considered abnormal with levels below 90% leading to potential long-term damage to organ systems. Cyanosis, or a physical bluish discoloration to the skin, mucous membranes, paws, tail or nose, is a physical cue often used to assess oxygenation and potential hypoxemia. Cyanosis happens well after internal damage is happening, around an SpO₂ reading of 80%6. Therefore, it is recognized that signs and symptoms of low SpO₂ are not reliable through physical examination of rodents using visible cues alone.
Preventing hypoxemia within research animals undergoing any sort of procedure, such as surgery or imaging, is done best by the use of a dedicated pulse oximeter. Pulse oximeters allow researchers to longitudinally assess SpO₂ within their research as well as maintain animal welfare during surgeries and to reduce variability by keeping vitals maintained and ensuring an appropriate anesthetic depth7-9.
Overview
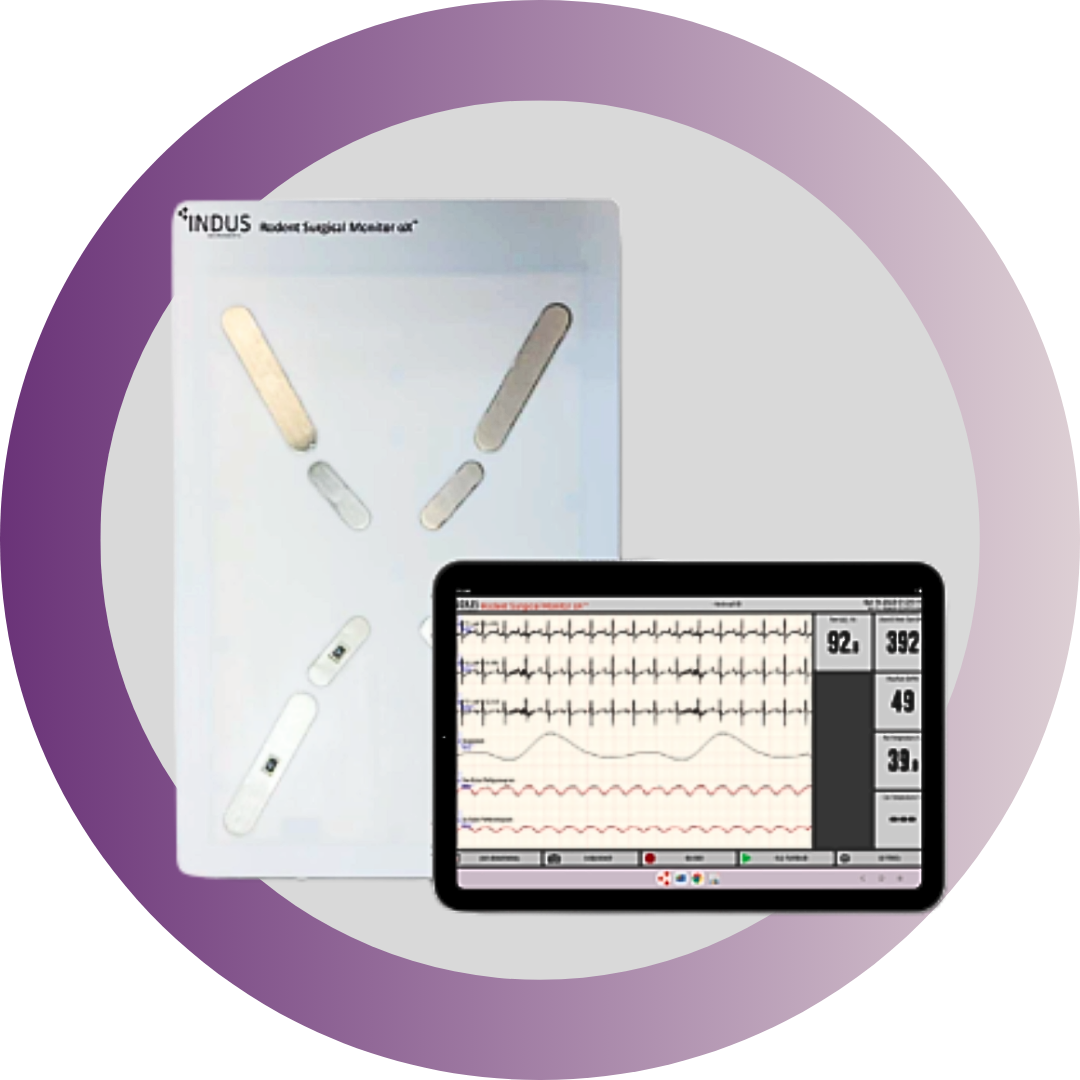
The white paper highlighted here uses a patent-pending oximeter built into the surgical platform that emits light pulses, both red and infrared light, and a photodetector to measure the SpO₂ within the paw of the rodent10. Most available pulse oximeters are large clips that are placed on the thigh, groin, ear or tail; of course, due to the small size of these animals, these clips are not optimized, leading to inaccuracies and variability during surgeries or experiments that directly affect oxygen saturation levels.
The sensor is sized down to be optimized for the rodents’ paw since the paw offers a reproducible site to continually monitor peripheral blood oxygenation noninvasively. The researchers highlight this new design that minimizes previous limitations and inaccuracies seen in other designs caused by movement of the pulse oximeter cable, caused by normal breathing of animal or when the technician making small movements during procedures.
Even when the animal is exposed to acute, severe hypoxemic episodes, this paw oximeter sensor shows less variability and drop-off of signal as compared to two other well-established sensors within the field, as highlighted in Figure 1.
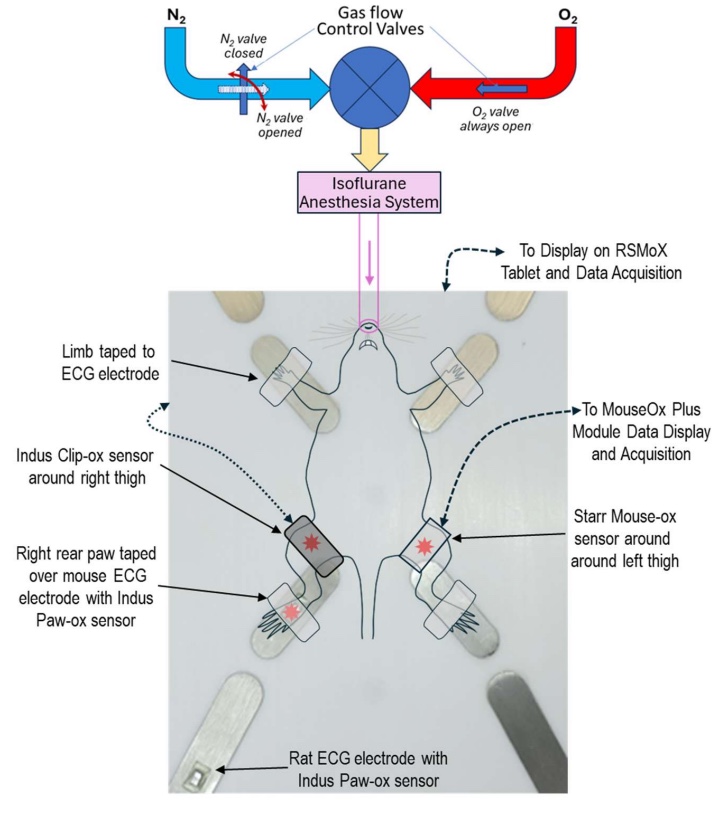
Figure 2 shows a schematic of a mouse placed on the platform highlighting the three pulse oximeter sensor placements. Along with SpO₂, the heated platform also integrates high-resolution ECG, respiration, core body temperature, and pressure (systemic and ventricular) monitoring to support improved animal welfare and more consistent experimental data. For a more detailed explanation of methodology and important findings.

Figure 1: Oxygen Saturation Curves During Hypoxic Events. Multiple SpO2 curves measured simultaneously by the three sensors over four severe, acute hypoxemic episodes in the 20-30 second.
Figure 2. Animal Placement on The Surgical Platform Highlighting Pulse Oximeter Placement. Experimental setup for pulse oximeter and other vital signs measurement in mice with RSMoX. The Indus Paw-ox and the Indus Clip-ox sensors are shown at the right rear paw and right thigh. The Starr Mouse-ox sensor is shown on the left thigh. *Figure and text adapted from Paw SpO₂ Measurement for Rodents Using Reflectance Pulse Oximeter. Indus Instruments. (2025)10.
Conclusion
Peripheral oxygen saturation (SpO₂) is becoming a more prevalent vital to monitor while working with research models like mice or rats, especially during procedures that require anesthesia since low oxygen levels can cause damage to organ and tissue systems. This white paper highlights a paw-optimized pulse oximeter designed specifically for rodents to accurately and reliably measure SpO₂.
References
- Vold, M. L., Aasebø, U.,Wilsgaard, T. & Melbye, H. Low oxygen saturation and mortality in an adult cohort: the Tromsø study.BMC Pulm. Med. 15, 9 (2015).
- Torp, K. D., Modi, P., Pollard, E. J. & Simon, L. V. Pulse Oximetry.JAMA298, 8724 (2023).
- Blood Oxygen Level: What It Is & HowToIncrease It. https://my.clevelandclinic.org/health/diagnostics/22447-blood-oxygen-level.
- Hafen, B. B. & Sharma, S. Oxygen Saturation. Oxygen Saturation -StatPearls- NCBI Bookshelf (nih.gov) 4–9 (2022).
- Bhutta, B. S.,Alghoula, F. & Berim, I. Hypoxia.StatPearls https://www.ncbi.nlm.nih.gov/books/NBK482316/ (2024).
- Snider, H. L. Cyanosis.Clinical Methods: The History, Physical, and Laboratory Examinationshttps://www.ncbi.nlm.nih.gov/books/NBK367/ (1990).
- Standards for Basic Anesthetic Monitoring. https://www.asahq.org/standards-and-practice-parameters/standards-for-basic-anesthetic-monitoring.
- Klein, A. A. et al. Recommendations for standards of monitoring during anaesthesia and recovery 2021: Guideline from the Association of Anaesthetists.Anaesthesia76, 1212–1223 (2021).
- Pulse Oximetry | Fact Sheets | Yale Medicine.https://www.yalemedicine.org/conditions/pulse-oximetry.
- Paw SpO2 Measurement for Rodents Using Reflectance Pulse Oximeter Method An Efficient and Reliable Method to Measure/Determine SpO2 in Rodents Rodent Surgical Monitor with Paw Pulse Oximeter Measurements. Indus Instruments. (2025).