Publication Highlight: Mineralocorticoid receptor antagonism prevents Coronary microvascular dysfunction in intermittent hypoxia independent of blood pressure
Importance
Obstructive sleep apnea (OSA) is a common disorder characterized by repeated obstruction to the airway during sleep. It is common for people with OSA to stop breathing for a few seconds in mild episodes; in severe cases, episodes can last 20 – 40 seconds and happen more than 100 times per hour1. This constant decrease or complete cessation of airflow results in intermittent hypoxemia, a low level of oxygen within the blood6. Intermittent hypoxemia creates a physiological stress response that will spike blood pressure and heart rate, thereby promoting inflammation and oxidative stress3. Over time, these chronic hypoxemic episodes place a substantial strain on the cardiovascular system, increasing the risk of hypertension, arrhythmia, and heart failure4,5. Contributing to heart failure, the coronary arteries that supply the heart with oxygen, can be damaged due to the intermittent hypoxemia6. Researching the mechanisms behind OSA as well as the comorbidities that can occur will be pivotal to advancing current therapeutics within this field.
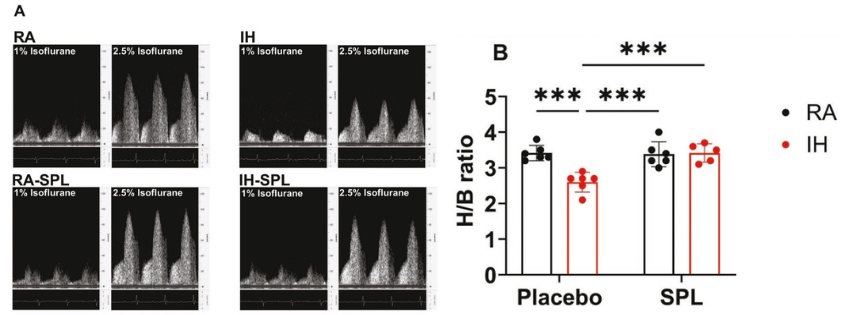
Figure 1. Spironolactone treatment improves CFVR in mice exposed to 6 weeks of IH. (A) Blood flow velocity images of coronary arteries responses at baseline (1%) and hyperemic inducing (2.5%) concentrations of isoflurane. (B) Coronary flow velocity reserve (CFVR) values are calculated as a ratio of hyperemic to baseline responses (H/B) to isoflurane. Data represented as mean ± S.D (n = 6) mice. Statistical analysis was done using two-way ANOVA followed by Tukey post hoc test. *** p <.001. IH, intermittent hypoxia; RA, room air; SPL, spironolactone. *Figure and text adapted from Badran, M. et al. Sleep. 2025.
Researchers from the University of Missouri recently demonstrated that coronary microvascular dysfunction associated with OSA can be targeted with the treatment of Spironolactone, a steroidal mineralocorticoid receptor inhibitor. The researchers used the Doppler Flow Velocity System to noninvasively measure coronary flow reserve, a key indicator of the coronary vessels’ capacity to dilate and increase blood flow to meet an increased metabolic demand to help prevent myocardial ischemia7,8. Spironolactone treatment restored coronary flow reserve in mice exposed to intermittent hypoxia for 6 weeks, a model mimicking OSA, as shown in Figure 19. The restoration of coronary flow reserve suggests improved dilation of the microcirculation supplying the heart as compared to the intermittent hypoxemia control group.

Figure 2. Hydrochlorothiazide treatment does not improve CFVR in mice exposed to 6 weeks of IH. (A) Blood flow velocity images of coronary arteries responses at baseline (1%) and hyperemic (2.5%) concentrations of isoflurane. (B) Coronary flow velocity reserve (CFVR) values are calculated as a ratio of hyperemic to baseline responses (H/B) to isoflurane. Data represented as mean + S.D (n = 5). Statistical analysis was done using two-way ANOVA followed by Tukey post hoc test. **p<.01,***p<.001,****p< .0001. IH, intermittent hypoxia; RA, room air; HTZ, hydrochiörothiazide. *Figure and text adapted from Badran, M. et al. Sleep. 2025.
Conclusion
Chronic kidney disease-dependent vascular complications were exacerbated when NF-κB pathway was suppressed, where IKK2 deficiency caused vascular calcification and stiffening leading to dramatically reduced survival within the IKK2 knockouts. Conversely, activating the NF- κB pathway by IκBα, inhibitor of pathway, deficiency significantly attenuated vascular calcification and stiffness through modulation of cell death, specifically by blocking cell death and reducing release of apoptotic calcifying–extracellular vesicle secretion.
References
- Chronic Kidney Disease (CKD): Symptoms & Treatment. https://my.clevelandclinic.org/health/diseases/15096-chronic-kidney-disease.
- Safar, M. E., London, G. M. & Plante, G. E. Arterial Stiffness and Kidney Function. Hypertension 43, 163–168 (2004).
- Boutouyrie, P., Chowienczyk, P., Humphrey, J. D. & Mitchell, G. F. Arterial Stiffness and Cardiovascular Risk in Hypertension. Circ. Res. 128, 864–886 (2021).
- Miyazaki-Anzai, S. et al. Activation of the IKK2/NF-κB pathway in VSMCs inhibits calcified vascular stiffness in CKD. JCI Insight 9, (2024).
- Zhang, J. et al. IκB kinase β (IKKβ): Structure, transduction mechanism, biological function, and discovery of its inhibitors. Int. J. Biol. Sci. 19, 4181 (2023).
- Wilkinson, I. B., Maki-Petaja, K. M. & Mitchell, G. F. Uses of Arterial Stiffness in Clinical Practice. Arterioscler. Thromb. Vasc. Biol. 40, 1063–1067 (2020).
- Hartley, C. J., Taffet, G. E., Michael, L. H., Pham, T. T. & Entman, M. L. Noninvasive determination of pulse-wave velocity in mice.
- Pilz, N. et al. Pulse Wave Velocity: Methodology, Clinical Applications, and Interplay with Heart Rate Variability. Rev. Cardiovasc. Med. 25, 266 (2024).
- Schwartz, J. E., Feig, P. U. & Izzo, J. L. Pulse Wave Velocities Derived from Cuff Ambulatory Pulse Wave Analysis: Effects of Age and Systolic Blood Pressure. Hypertension 74, 111–116 (2019).
- Najjar, S. S. et al. Pulse Wave Velocity Is an Independent Predictor of the Longitudinal Increase in Systolic Blood Pressure and of Incident Hypertension in the Baltimore Longitudinal Study of Aging. J. Am. Coll. Cardiol. 51, 1377 (2008).