Publication Highlight: Activation of the IKK2/NF-κB pathway in VSMCs inhibits calcified vascular stiffness in Chronic Kidney Disease
Importance
Chronic kidney disease occurs with progressive damage to the kidneys with loss of their ability to properly filter waste products1. Vascular stiffness can directly further promote structural damage to the kidneys, as well as increase blood pressure and increase cardiac workload2,3. Over half of the deaths from this diagnosis are due to cardiovascular disease so interplay between the kidneys, the vascular system, and the heart are of importance to research. Researchers from the University of Colorado Anschutz Medical Campus recently showed that activation of the IKK2/NF-κB inflammatory pathway in vascular smooth muscle cells plays a protective role in chronic kidney disease-dependent calcified vascular stiffness4.
Overview
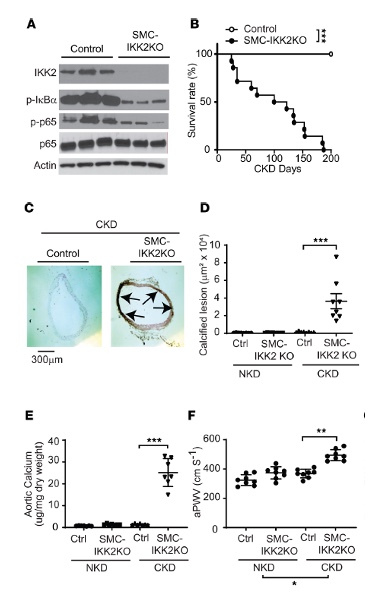
Chronic kidney disease induces NF-κB–mediated inflammation by IKK2 and this pathway was thought to exacerbate the disease progression5. Using a chronic kidney disease model with IKK2 deficiency in the aortic media caused calcified arterial calcification and stiffness, as highlighted in Figure 1. In brief: A) validation of the tamoxifen-induced IKK2 knockout as well as reduction in the subsequent downstream signaling proteins, p-IκBα and p-p65. B) reduction in lifespan of the animal. C-D) Histological analysis of the dissected aortic arch highlighting calcified deposits as well as E) increased aortic calcium content, indicating calcification. F) Using the Doppler Flow Velocity System, the group used two probes simultaneously to determine how quickly the pulse of blood was arriving from one site near the heart, transverse aortic arch, to another site lower in the periphery, abdominal aorta. Of note, Pulse wave velocity is the gold standard, non-invasive measurement for arterial stiffness and vascular aging both clinically and within research6–8 and an independent predictor of hypertension, cardiovascular events and mortality9,10. Here the researchers confirmed aortic stiffening within the IKK2 knockout.
Figure 1. VSMC IKK2 deficiency induces early mortality and calcified vascular stiffness in mice with CKD. (A) Immunoblot analysis of the IKK2/NF-κB pathway in the aortic media of SMC-IKK2–KO mice. Eight-week-old control and SMC-IKK2–KO mice (n = 8) were intraperitoneally injected with tamoxifen for 5 days and subjected to 5/6 nephrectomy (CKD). Three weeks after the surgeries, animals were euthanized. (B) Survival rate of SMC-IKK2–KO mice under CKD. (C) Histological and (D) quantitative analysis of aortic arches with von Kossa staining. Aortas were dissected from the mice 3 weeks after CKD was induced. Arrows (black) indicate calcified lesions. Scale bar: 300 μm. (E) Aortic calcium content. Aortic calcium content was analyzed with an ash assay coupled with a colorimetric calcium assay. (F) aPWV was analyzed using an Indus Doppler Flow Velocity System 3 weeks after CKD was induced.*Figure and text adapted to highlight the vasculature morphological findings from Miyazaki-Anzai, S. et al. JCI Insight. 20244.
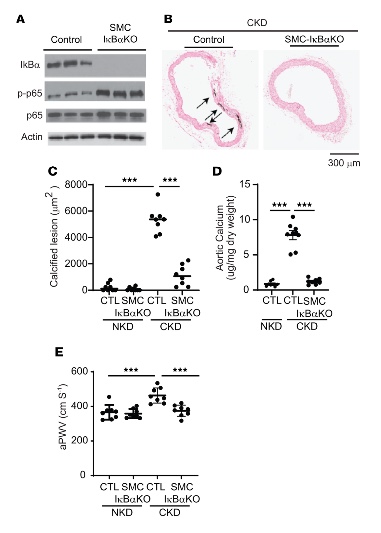
Since inactivation of the NF-κB pathway in the chronic kidney disease model caused calcified aortic stiffening and lethality, the researchers then investigated the effect of activation of NF-κB pathway on chronic kidney disease. Knocking out of IκBα, a key inhibitor of the NF-κB pathway, in the aortic media is protective to vascular smooth muscle cells and within chronic kidney disease, as highlighted in Figure 2. In brief: A) validation of the tamoxifen-induced IκBα knockout as well as subsequent increase in an activated NF-κB downstream subunit, p-p65. Of note, there was no significant reduction in lifespan within these knockouts and B-C) histological analysis of the dissected aortic arch when the chronic kidney disease controls started to develop pathology and calcified deposits, while this was mediated in the IκBα knockout as well as D) decreased aortic calcium content as compared to controls. E) an increased pulse wave velocity within the controls with chronic kidney disease pathology is attenuated within the IκBα knockout group.
Figure 2. VSMC IκB deficiency attenuates CKD-induced calcified vascular stiffness. (A) Immunoblot analysis of the IKK2/NF-κB pathway in the aortic media of SMC-IκBα–KO mice. Eight-week-old control and SMC-IκBα–KO mice (n = 8) were intraperitoneally injected with tamoxifen for 5 days and subjected to 5/6 nephrectomy (CKD). Twelve weeks after the surgeries, animals were euthanized. (B and C) Histological analysis of aortic arches with von Kossa staining. Aortas were dissected from the mice 12 weeks after sham or 5/6 nephrectomy operations. Arrows (black) indicate calcified lesions. Scale bar: 300 μm. (D) Aortic calcium content. Aortic calcium content was analyzed with an ash assay coupled with a colorimetric calcium assay. (E) aPWV was analyzed using an Indus Doppler Flow Velocity System 12 weeks after CKD was induced. *Figure and text adapted to highlight the vasculature morphological findings from Miyazaki-Anzai, S. et al. JCI Insight. 20244.
Conclusion
Chronic kidney disease-dependent vascular complications were exacerbated when NF-κB pathway was suppressed, where IKK2 deficiency caused vascular calcification and stiffening leading to dramatically reduced survival within the IKK2 knockouts. Conversely, activating the NF- κB pathway by IκBα, inhibitor of pathway, deficiency significantly attenuated vascular calcification and stiffness through modulation of cell death, specifically by blocking cell death and reducing release of apoptotic calcifying–extracellular vesicle secretion.
References
- Chronic Kidney Disease (CKD): Symptoms & Treatment. https://my.clevelandclinic.org/health/diseases/15096-chronic-kidney-disease.
- Safar, M. E., London, G. M. & Plante, G. E. Arterial Stiffness and Kidney Function. Hypertension 43, 163–168 (2004).
- Boutouyrie, P., Chowienczyk, P., Humphrey, J. D. & Mitchell, G. F. Arterial Stiffness and Cardiovascular Risk in Hypertension. Circ. Res. 128, 864–886 (2021).
- Miyazaki-Anzai, S. et al. Activation of the IKK2/NF-κB pathway in VSMCs inhibits calcified vascular stiffness in CKD. JCI Insight 9, (2024).
- Zhang, J. et al. IκB kinase β (IKKβ): Structure, transduction mechanism, biological function, and discovery of its inhibitors. Int. J. Biol. Sci. 19, 4181 (2023).
- Wilkinson, I. B., Maki-Petaja, K. M. & Mitchell, G. F. Uses of Arterial Stiffness in Clinical Practice. Arterioscler. Thromb. Vasc. Biol. 40, 1063–1067 (2020).
- Hartley, C. J., Taffet, G. E., Michael, L. H., Pham, T. T. & Entman, M. L. Noninvasive determination of pulse-wave velocity in mice.
- Pilz, N. et al. Pulse Wave Velocity: Methodology, Clinical Applications, and Interplay with Heart Rate Variability. Rev. Cardiovasc. Med. 25, 266 (2024).
- Schwartz, J. E., Feig, P. U. & Izzo, J. L. Pulse Wave Velocities Derived from Cuff Ambulatory Pulse Wave Analysis: Effects of Age and Systolic Blood Pressure. Hypertension 74, 111–116 (2019).
- Najjar, S. S. et al. Pulse Wave Velocity Is an Independent Predictor of the Longitudinal Increase in Systolic Blood Pressure and of Incident Hypertension in the Baltimore Longitudinal Study of Aging. J. Am. Coll. Cardiol. 51, 1377 (2008).