Modality Review: Introduction to Intravital Microscopy
When to Consider Using Intravital Microscopy
Intravital Microscopy (IVM) provides real-time imaging of cellular events occurring within an intact imaging subject; providing a comprehensive picture of a wide variety of dynamic processes. Recent advancements in microscopic techniques, as well as fluorescent tagging techniques has permitted imaging of deeper tissues with higher resolution on a wider variety of cell types and imaging targets. IVM is a vital technique used in tissue engineering, regenerative medicine, and examining host reactions to implanted biomaterials. Additionally, IVM has been used by researchers in almost every disease area, including cancer biology, neurology, etc.
IVM allows for cellular events to be imaged in their native environment in real-time; with the advent of imaging windows and chambers, these complex and dynamic processes may be observed over the course of longitudinal study. This differs from postmortem histological examination of fixed tissues, in which cellular events may be examined, but only at a fixed moment in time. Cellular events such as proliferation, migration, differentiation, etc. may be imaged along with specific cell-cell interactions, as examples.
Many of the imaging modalities discussed so far in this series have relevance both preclinically and clinically, however, due to the invasive nature of intravital microscopy this modality is not used in the clinic. The findings and outcomes discovered using intravital microscopy are however very relevant to the clinic and have led to many advancements in treating a wide variety of clinical conditions.
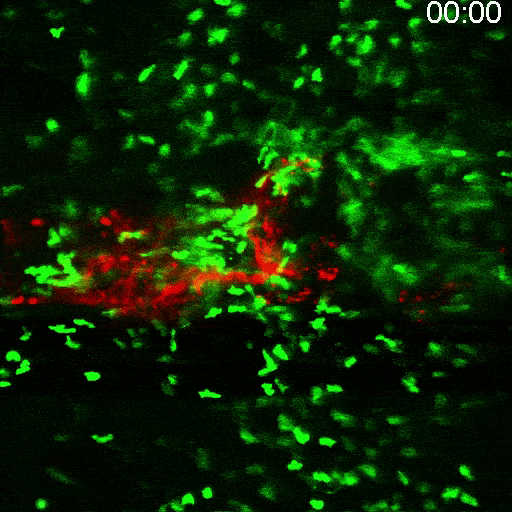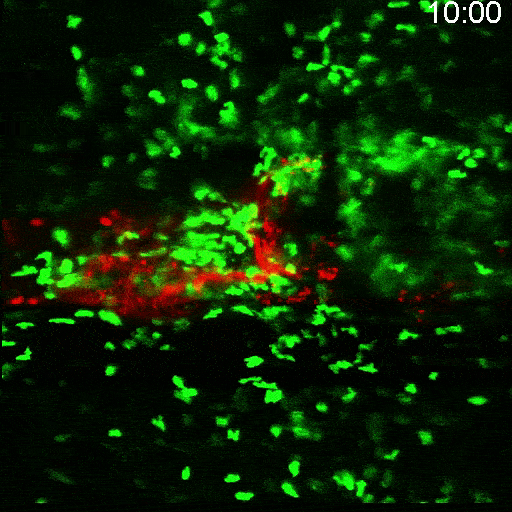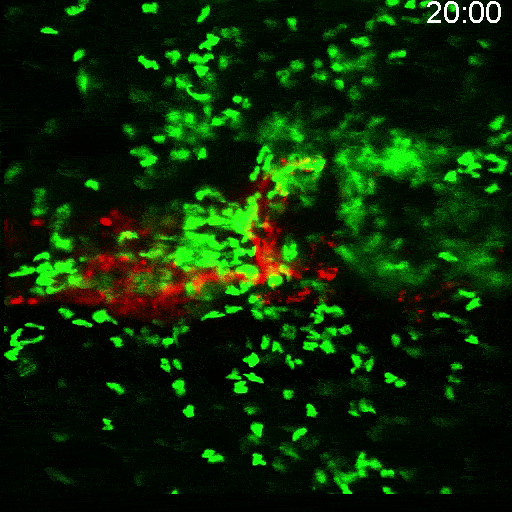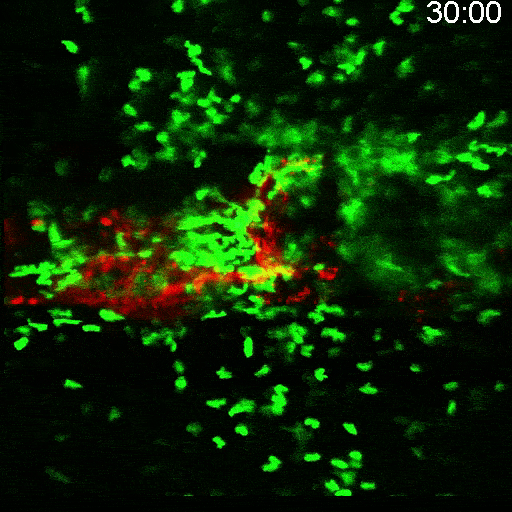
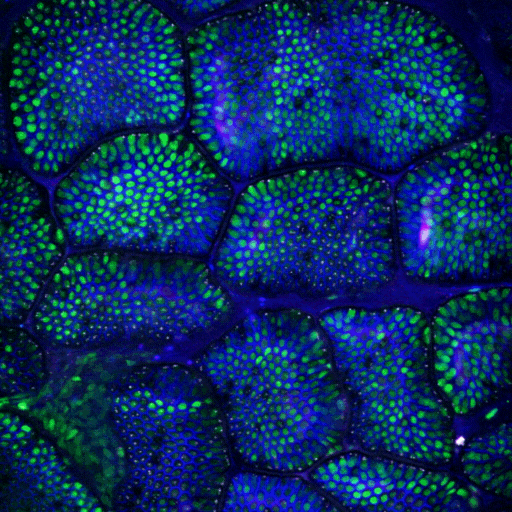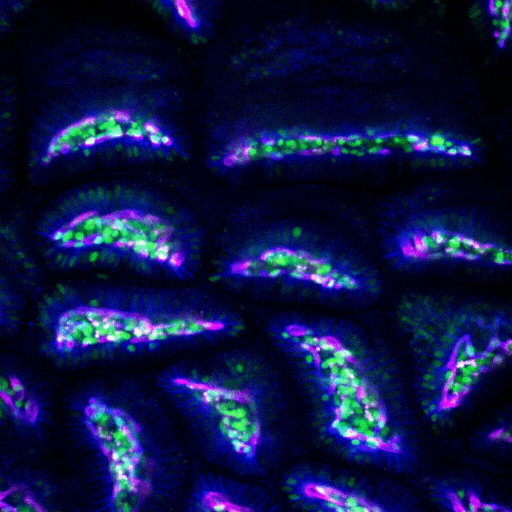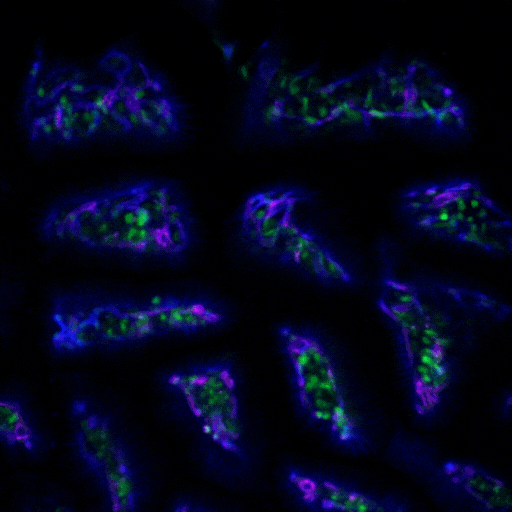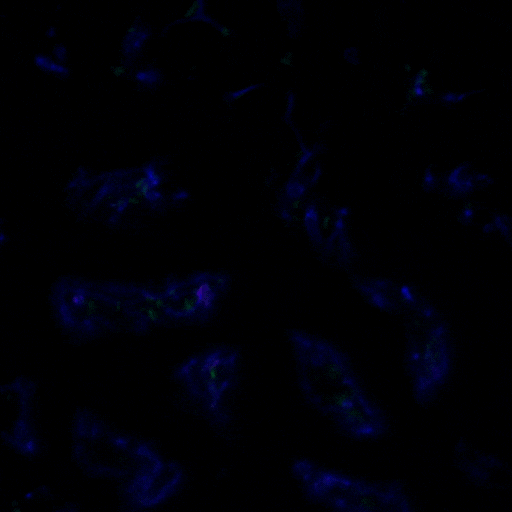
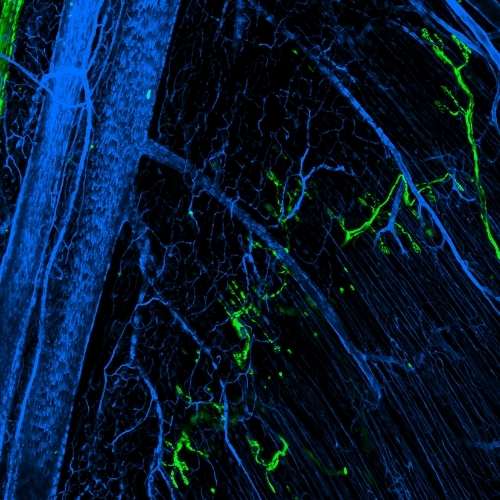
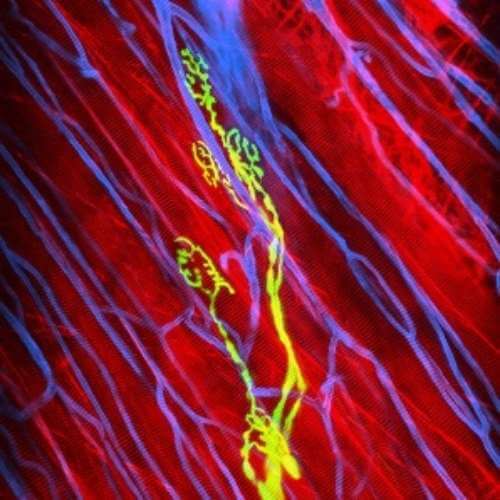
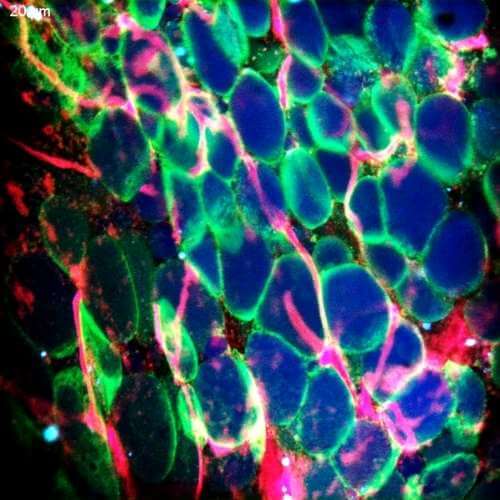
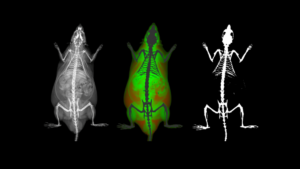
Multimodal Imaging
Intravital microscopy is at its most basic definition multimodal, in that within a single study multiple biomarker, or imaging targets can be observed simultaneously. Taken a step further, IVM is complimentary to whole body imaging modalities in that it is both important to understand a biological or disease process on the whole body, or organ level, but further elucidation of what is going on at the microscopic cellular level is also vitally important to the overall understanding of a specific disease or therapeutic response.
Fully Integrated Intravital Microscopy System
The IVM systems, manufactured by IVIM Technologies and distributed by Scintica, have been designed as a fully integrated intravital microscopy system for preclinical imaging applications. Specifically, the IVM system are an all-in-one design allowing researchers to quickly and easily perform confocal and/or two-photon intravital microscopy studies on small animal imaging subjects, such as mice. The system includes a heated stage, integrated anesthesia (if desired), and physiological monitoring to ensure the welfare of the animal throughout the imaging session. In many labs, integrating all of this onto a conventional confocal or two-photon microscope can take an extended period of time, and leads to a challenging animal set-up. Additionally, a wide variety of optical windows and chambers are available so that researchers new to the field are able to again start optimizing their animal model without spending time optimizing the equipment set-up.
Finally, the IVM systems are modular in design allowing for an optimized configuration to be easily selected, and expanded upon in the future if needed. The software interface is easy to use, again allowing those new to the field to start optimizing their own models quickly, without spending time on learning how to use the software.
There are some advanced features like motion correction which result in drastically improved images – for example in the lung, abdomen, and heart; as well as ultrafast scanning to track the movement of several cells over time. Finally, there is also the ability to perform both 3D Z-stack images, as well as 4D image rendering.
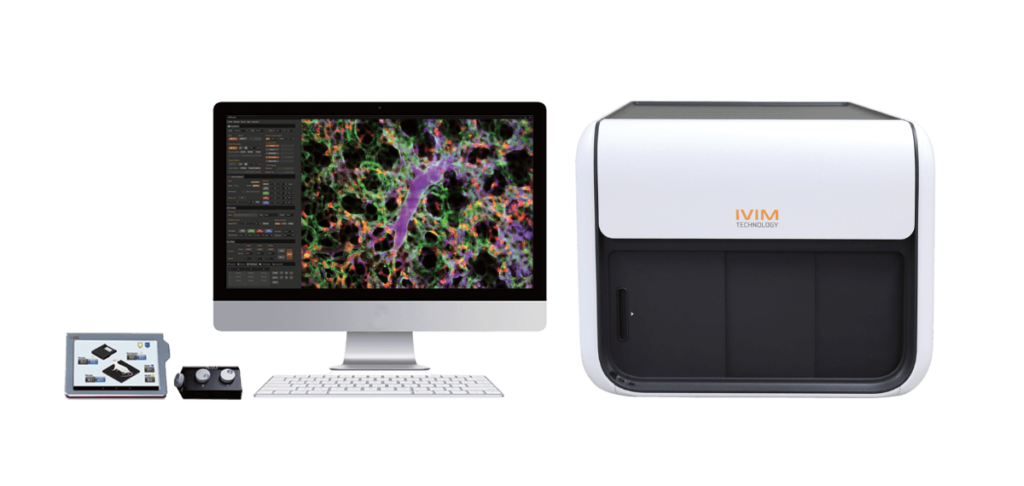
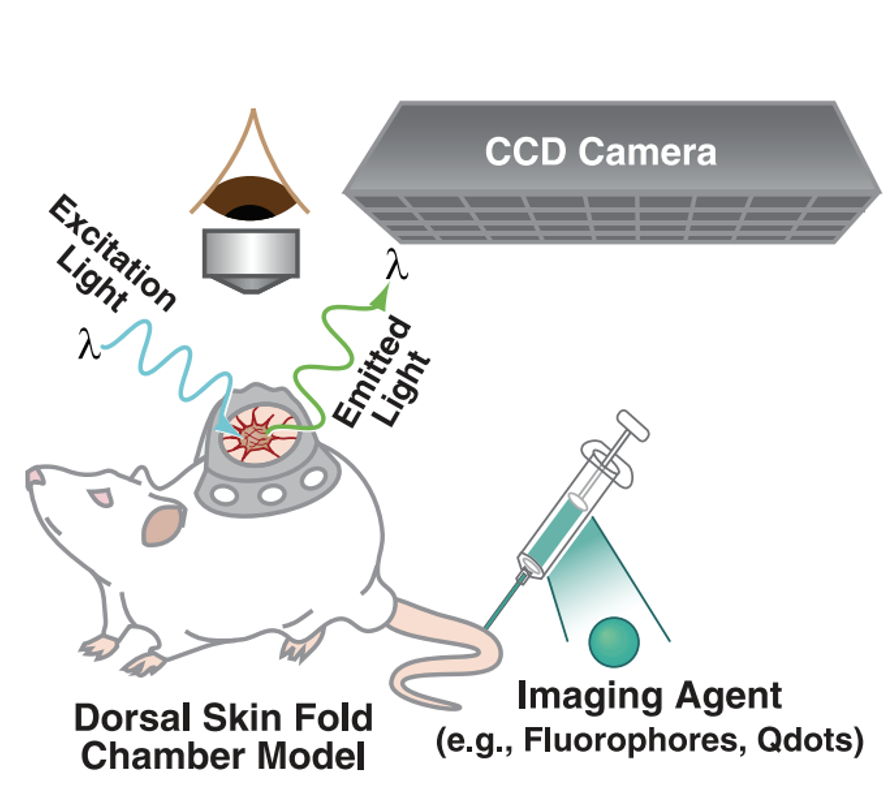
Figure adapted from – James ML, Gambhir SS. Physiological reviews. 2012
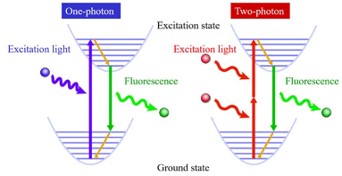
Figure adapted from –
https://www.frontiersin.org/articles
/10.3389/fchem.2020.617463/full; 2023
Basic Explanation of How Intravital Microscopy Works
The development of intravital microscopy was dependent on a few key advancements, including the development of a fluorescence microscope in the very early 1900’s, further to that was the need for exogenous fluorophores, and also the development of the confocal scanning microscope much later in the 20th century. The development of multiphoton microscopy to increase the dept of penetration which was possible without the risk of phototoxicity. Finally, the more recent advancements of optical windows and chambers in the 21st century has further increased the utility of intravital microscopy in vivo.
Confocal microscopy was developed as a means of limiting the detected signal to a specified focal plane. This is accomplished by focusing the excitation light onto a single point, and a pinhole aperture on the optics cuts away light originating outside of the focal spot.
Two-photon microscopy was further developed to help improve resolution. In this situation two photons of approximately the single-photon wavelength interact with a fluorescent molecule within a very short time span. The light is again focused on a specific spot, and the changes of the two photons interacting with the molecule simultaneously is limited to a small area. Three dimensional images are accomplished by scanning the focal spot over and through the specimen.
While the optical techniques needed for intravital microscopy are described above, of equal importance is the ability to differentiate between different tissue and cell types. This can be achieved by application of a targeted fluorescent dye; by the injection of fluorescently labelled cells, nanoparticles, small molecules, or antibodies; or by the use of genetic reporters which create a fluorescent label under specific controlled or activatable conditions.
For chronic studies, a specialized optical window or chamber must be surgically implanted over the area/organ of interest. Alternatively, acute studies may be performed by surgically exposing the area/organ of interest, however these studies are shorter and most of the time are terminal studies.
Our Next Modules
Preclinical Imaging Modalities
Available Now
As we begin to explore the idea of multimodal imaging, let’s first start to explore the variety of preclinical imaging modalities that are most commonly used by researchers around the world.
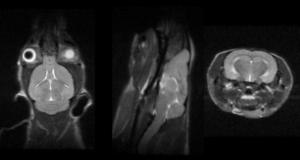
Modality: MRI
Available Now
MRI is considered the gold standard in soft tissue imaging, both in the clinic on patients and by researchers on a wide variety of preclinical imaging subjects.
Modality: CT
Available Now
Computed Tomography (CT) is one of the most commonly used clinical imaging techniques, next to perhaps ultrasound.
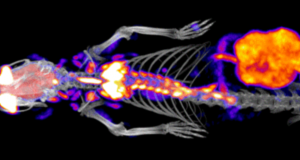
Modality: PET
Available Now
Nuclear imaging modalities include positron emission tomography (PET) as well as single photon emission computed tomography (SPECT), which both detect gamma radiation emitted from a radionucleotide injected into an imaging subject.
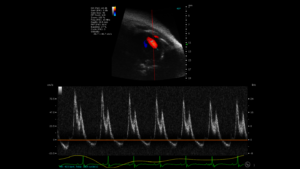
Modality: Ultrasound
Available Now
Ultrasound (US) imaging is one of the most commonly used diagnostic techniques clinically, and is widely used in preclinical imaging as well. It is a safe, non-invasive, and relatively inexpensive technology compared to some of the other available imaging modalities.
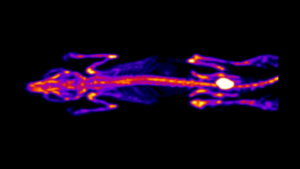
Modality: Optical
Available Soon
When discussing Optical Imaging techniques in this series, the meaning is to cover bioluminescence (BLI) and fluorescence (FLI) imaging. T
Modality: Photoacoustic
Available Now
Photoacoustic (PAI) imaging is a technology which combines the sensitivity of optical imaging with the depth of penetration and resolution of ultrasound imaging.
Modality: Microscopy
Available Now