How Theranostics Changed the Way We Diagnose and Treat Children with Neuroblastoma
Each year, between 700 to 800 children are diagnosed with neuroblastoma in North America, 90% of them under the age of 5. Around half of the children are considered at high risk of becoming refractory to, or relapsing following intensive and multimodal therapy (chemotherapy, surgery, and radiotherapy). Sadly of the children who relapse, only 10% will live beyond 10 years. Neuroblastoma still accounts for 15% of children’s cancer death.
There is an unmet need for smarter and safer treatments that are tailored to the molecular, pathologic, or radiologic tumor characteristics of each child – a.k.a. Precision medicine. A small molecule or antibodies that can be used to both diagnose and treat, represent one invaluable strategy, termed Theranostics, to deliver precision medicine to cancer patients. Children with neuroblastoma were among the first to benefit from theranostics, with radiolabeled meta-iodo-benzyl-guanidine (mIBG).
Neuroblastoma arises as a defect of embryonic development of the nerve cells of the sympathetic nervous system, and can thus develop anywhere along the system often starting in the abdomen and adrenal glands (30%). Asymptomatic, children with neuroblastoma often present at the time of diagnosis with a huge abdominal mass that encompasses major blood vessels, making surgery difficult, without prior debulking of the tumor mass with chemotherapy. At diagnosis, half of the patients present metastatic disease, most commonly in the bone and marrow. While MRI and CT represented useful imaging modalities for diagnosis, response assessment, and planning of the surgical resection of the primary tumor, mIBG-based scintigraphy represented a pivotal addition to the arsenal of diagnostic tools to assess whole-body disease, surpassing standard-of-care bone scan (Technetium-99m) in both sensitivity and specificity.
Neuroblastoma almost exclusively expresses the norepinephrine transporter (NET). mIBG is a norepinephrine analog, and as a result, its uptake is a sensitive and specific marker for neuroblastoma and a reporter for molecular imaging-based detection of neuroblastoma with planar scintigraphy or single-photon emission computerized tomography-computed (SPECT) when labeled with 123-I or more recently with positron emitting tomography when labeled with 124-I. 123-I MIBG scans are now a ubiquitous component of the staging, response assessment, and surveillance for children with neuroblastoma as illustrated below by 123I-mIBG scans of an 8½ -year-old girl with widespread skeletal involvement of stage 4 neuroblastoma, which was free of disease after induction therapy and surgery before unfortunately relapsing.
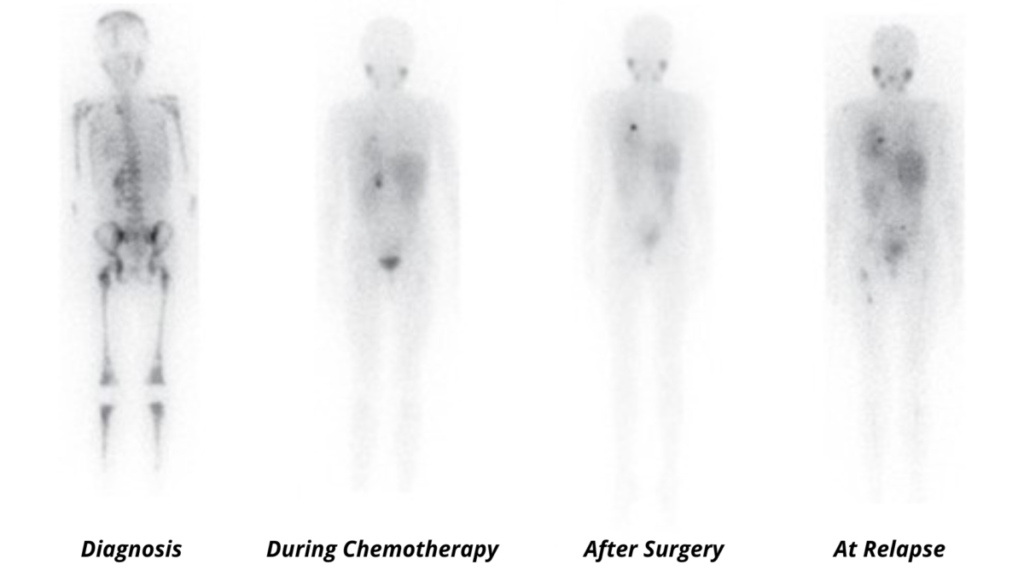
Adapted from Matthay KK et al., Brit. J. Cancer. (2010) 102:1319–1326
When labeled with 131-I, mIBG becomes a therapeutic radiopharmaceutical agent that can be administered systemically, to deliver targeted radiotherapy to neuroblastoma cells. 131-I mIBG therapy has shown efficacy in children with newly diagnosed neuroblastoma and those with relapsed disease and continues to be investigated in combination with chemotherapy and immunotherapy.
In summary, the success of theranostics in neuroblastoma is a paradigm that demonstrates the potential role of molecular imaging and theranostics to guide the delivery of smarter and better treatments for all children with cancer. Much more needs to be done to find a cure for neuroblastoma and other pediatric cancer. During September, you can help raise awareness of childhood cancer by simply sharing articles, tweets, and experiences that resonate with you!
Author
Tyler Lalonde, Ph.D – Product Manager – Nuclear Medicine – Scintica