(March 14, 2024) Webinar: Validation of DEXA for Longitudinal Quantification of Tumor Burden in A Murine Model of Pancreatic Ductal Adenocarcinoma
Noninvasive imaging is central to preclinical, in vivo models of pancreatic ductal adenocarcinoma (PDAC). While bioluminescent imaging (BLI) is a gold standard, its signal is dependent on the metabolic activity of tumor cells. In contrast, dual energy X-ray absorptiometry (DEXA) is a direct measure of body composition. Thus, this project aimed to assess the potential of using DEXA for longitudinal quantification of tumor burden versus BLI in an orthotopic KCKO murine model of PDAC. In short, DEXA successfully identified a growing tumor burden and accurately predicts ex vivo tumor mass in a time sensitive manner.
Learning objectives:
- Learn to take advantage of DEXA for things other than bone density and bone health (i.e., lean, and fat mass)
- Understand that DEXA can reproducibly and accurately be used to monitor tumor burden and growth in orthotopic murine models of pancreatic cancer
- Understand the importance of repurposing techniques and equipment for new analysis
- Understand that non-invasive in vivo imaging is crucially important in severely compromised models like those for PDAC and other cancers
- See the value of utilizing multiple techniques throughout an experiment to enhance data collection
Overview:
Pancreatic ductal adenocarcinoma (PDAC) is a highly malignant cancer currently responsible for 8% of cancer related deaths in the United States. Despite continued advances and an in-depth understanding of its pathology, the 5-year survival rate remains at a dismal 12%, the lowest among all cancers. Preclinical models of PDAC are essential tools that researchers use to investigate mechanisms of tumor initiation, engraftment, and progression, as well as efficacy of novel therapies. These studies require reliable in vivo imaging techniques that accurately access tumor growth and response to treatment. While numerous imaging modalities have been utilized for preclinical cancer research, each has its unique strengths and limitations.
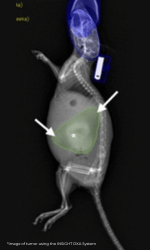
Bioluminescent imaging (BLI) has been widely adopted as a gold standard due to its relatively low cost and impressive sensitivity. BLI relies on de novo transgenic expression of firefly luciferase and injection of its luciferin substrate, which produces light that can be detected and quantified as photon/second/cm2/steradian (p/s/cm2/sr) via a CCD camera system. As such, a major disadvantage of this approach is the requirement of genetically modified tumor models. Furthermore, as BLI signal intensity is a function of tumor metabolism rather than tumor size, it must be used in conjunction with another outcome measure that directly assesses tumor mass. Thus far, the focus in preclinical models of PDAC has been ultrasound and magnetic resonance imaging (MRI), which have sensitivity and specificity strengths, but suffer from high costs at low throughput. To overcome this, the lab developed a dual energy X-ray absorptiometry (DEXA) approach to quantify PDAC-induced skeletal muscle wasting using a non-metastatic model of PDAC. To expand on this success, they investigated DEXA’s ability to detect the engraftment of orthotopically implanted luciferase expressing PDAC cells (KCKO-Luc), tumor growth over time, and accuracy in predicting ex vivo tumor mass versus BLI, in our murine model.
It was observed in their lab that not only is BLI an inconsistent and inaccurate measure of tumor burden over time, but also find that increased mouse handling and constant anesthetization further exacerbates a decline in health at late stages of disease. Thus, the present study aimed to adapt DEXA for the reliable and reproducible quantification of tumor burden to reduce the stress placed on these animals. In doing so, it reduces potential unwanted health effects and improve experimental reproducibility. The lab found that DEXA reliably identifies a tumor burden starting at week 2 post inoculation when compared to non-tumor control mice (NTC) and successfully quantifies and increasing burden over the course of 10 weeks. Furthermore, they showed that DEXA quantification correlates strongly with ex vivo tumor weight and provides reproducible measures when analyzed by independent observers, validating it as a reliable method for longitudinal tumor quantification.
They concluded from this study, that DEXA is a rigorous method for quantifying tumor burden in vivo but there are distinct advantages and disadvantages for both BLI and DEXA and these were thoroughly described in the presentation. Zachary Sechrist highlighted the previously published data that supports these claims as well as make an argument for DEXA in other preclinical cancer models like xenograft and transgenic models. Lastly, he acknowledged how they now use both BLI and DEXA moving forward to obtain an enhanced understanding of tumor development in this model and noted how this compares to other accepted modalities like PET, MRI and CT imaging.
About the Speaker (s)

Zach Sechrist,
3rd year PhD student at the University of Rochester School of Medicine and Dentistry in the Pathology and Laboratory Medicine
Zach is currently a 3rd year PhD student at the University of Rochester School of Medicine and Dentistry in the Pathology and Laboratory Medicine program. He has always had an interest in science, and it wasn’t until his mother was diagnosed with stage II breast cancer that he developed a passion for biomedical research. Seeing how hard the diagnosis is on a patient and their family, he sought to pursue a career with the potential to reduce this burden for others. Zach was first introduced to research as an undergraduate student where he began working on a project investigating biomarkers for patients with fragile X syndrome and this is where he developed an interest in lab work. To strengthen his laboratory skills and gain experience in industry research, he accepted an intern position at Regeneron Pharmaceuticals for the summer of 2019. His project involved optimizing protein clean up protocols and subsequent western blotting protocols with the goal of validating the purity of experimental antibody drugs. These experiences led him to continue his research career and apply for graduate school.
After his acceptance into University of Rochester School of Medicine and Dentistry, it was an easy decision based on the exceptional research and resources available. Zach was given the opportunity to join Dr. Calvin Cole’s laboratory in summer 2022. HIs work to this point has been investigating the molecular mechanisms underlying pancreatic cancer induced skeletal muscle wasting. Moving forward, he currently is working to investigate the role of insulin-like growth factor binding protein (IGFBP-3) on disease progression and develop and characterize a monoclonal antibody therapeutic against IGFBP-3. His plans after graduate school involve pursuing a post-doctoral fellowship and entering academia as a PI of his own research laboratory.
