System Used:
SuperArgus PET/CT

The endocannabinoid system (ECS) is not a body transmission system that everyone has heard about, however, it plays a crucial role in everyday bodily functions. Some of the functions it controls are learning and memory, emotional processing, sleep, temperature control, pain control, inflammatory and immune responses, and eating1 . The system is comprised of an entire network of chemical entities and cell receptors that are spread throughout the brain and the rest of the body. The largest of these receptors is the cannabinoids found in the brain and these are stimulated by molecules called the endocannabinoids. When they interact with the cannabinoid receptors, they will trigger immediate feedback for functions such as hunger and temperature control1.
Two of the main and more well-studied endocannabinoids are N-Arachidonoylethanolamine (AEA) and 2-arachidonoylglycerol (2-AG). AEA binds and activates both the cannabinoid 1 and 2 receptors as a partial agonist (partial activation). 2-AG will bind to both receptors as well; however, it will cause full activation of the receptors2. However, there are enzymes that can break down AEA and 2-AG. Fatty acid amine hydrolase (FAAH) breaks down AEA, while monoacylglycerol (MAGL) breaks down 2-AG. When these enzymes are activated, there is an inactivation of endocannabinoid signaling leading to the production of arachidonic acid (AA). Inhibitors of FAAH and MAGL have been investigated for the treatment of various human diseases such as pain and neuroinflammation3. Because there are scaffold inhibitors for both these enzymes it is possible to alter their structures to generate usable positron emission tomography (PET) agents to image conditions such as neuroinflammation.
The goal of He, Y, and coworkers was to use the previous scaffold of MAGL inhibitors and modified these structures to include carbon-11 (11C) to generate useable reversible PET tracers to tract MAGL for applications in monitoring neuroinflammation4. The authors first started with 617 compounds and used them in silico experiments with multiparameter optimization (MPO) to narrow down lead candidates that would have the best potential to bind to MAGL reversibly. Two structures with MPO scores of 3.8 and 4.1 were identified. This was followed up by a passive membrane permeability assay (PAMPA), lipid membrane binding assay (LIMBA logD), and plasma membrane binding assay to predict non-specific binding (NSB). These results were conclusive that the two hit compounds would both have high on-target binding, and high brain permeability4. These two compounds were then labeled with 11C via 11CO2 fixation. A MAGL knockout (KO) and wild-type (WT) mouse model were used to measure in vivo specificity and selectivity followed by in vitro autoradiography on excised brain tissue4. The SuperArgus PET/CT scanner was used for in vivo experiments. The two lead compounds can be visualized in Figure 1 below.
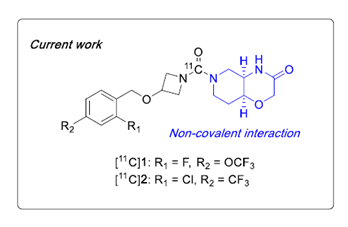
Figure 1. Structure of top two MAGL inhibitors identified from in silico experiments and in vitro binding/permeability experiments.
Brain tissues were obtained from C57/B16 mice and Wistar rats and cut to a 10 µm thickness and then subjected to the two 11C tracers see in Figure 1. Each tracer was blocked via a non-radioactive compound at a concentration of 10 µM. Autoradiograms can be visualized in Figure 2 below4.
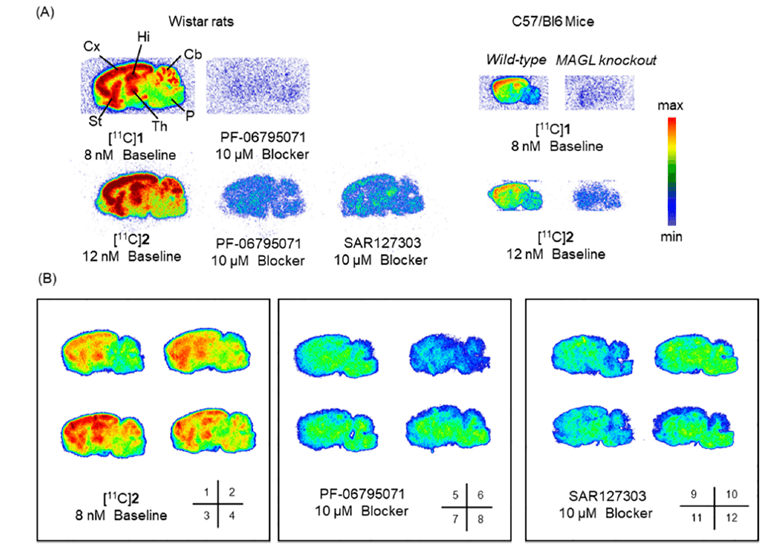
Figure 2. Representative autoradiograms of the ex vivo tissue in both rat and mice brain tissues. Rat brains were blocked via non-radioactive tracers (PF-06795071 and SAR127303) and a MAGL knockout mice model was also used.
Based on the images above both tracers showed both specificity and selectivity in both rats and mice. Higher levels of tracer accumulation were identified in the cortex, striatum, hippocampus, and thalamus of the rat, while moderate accumulation of the tracers was observed in the cerebellum. This signal was blocked by non-radioactive tracers (PF-06795071 and SAR127303). Similar observation was seen in the mouse brains, however the control used was a MAGL KO model. There was significant uptake in the WT with little to no signal in the KO model again highlighting the specificity and selectivity of both these tracers. Panel B in Figure 2 is the same experiment in the rat brain but prior to imaging with radioactive and non-radioactive blocking tracers washing was not performed. This was done to better mimic the in vivo situation4.
In vivo experiments were performed with the SuperArgus PET/CT scanner on MAGL KO and WT mice. This was done to measure whole brain activity time curves (TACs). However, based on the in vitro and ex vivo results compound 2 in Figure 1 was superior in both specificity and selectivity and therefore compound 2 was the only one assessed the in vivo mouse model. SUV were lower in MAGL KO mice compared to WT indicating specific binding of compound 2 in vivo. To assess if compound 2 is a P-glycoprotein (P-gp) a P-gp inhibitor was used in combination with the 11C tracer. The higher SUV at 1 min post-injection (p.i.) highlighted compound 2 acting as a P-gp4. These results are summarized in Figure 3 below.
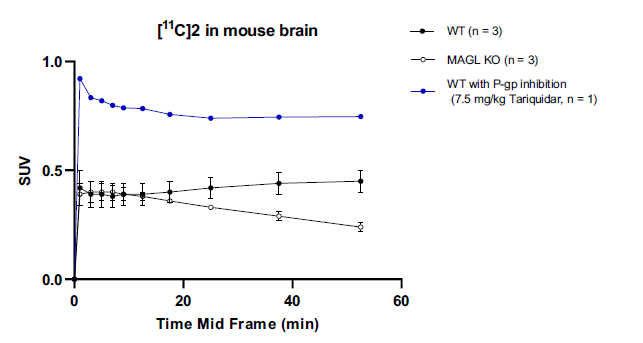
Figure 3. TACs for whole brain accumulation of compound 2 in the brain of a MAGL KO mouse model.
Radiometabolite analysis was performed using ultra-pressure liquid chromatography (UPLC) with acetonitrile extracts from the brain, plasma, and urine. Greater then 95% of the radioactivity in the brain was that of the intact tracer, and 33% was intact within the plasma. It was also the dominant species in the urine. This concludes that the signal acquired from the brain is the intact molecular probe. These results are summarized in Figure 4 below4.
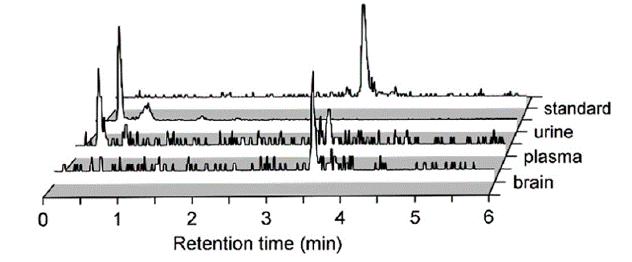
Figure 4. Radiometabolite analysis of compound 2 in the brain, plasma, and urine using UPLC.
In conclusion, the authors were able to identify a lead 11C MAGL-specific tracer from a group of over 600 compounds by starting with in silico experiments, and then moving into in vitro and in vivo analysis of the lead compound 2. Compound 2 had high selectivity and specificity for MAGL in the brain along with great stability in vivo. This compound can be used as a non-invasive imaging tool to follow the progression of MAGL in conditions such as neuroinflammation. The SuperArgus PET/CT scanner was a useful tool in coming to this conclusion by being able to determine the high level of sensitivity and specificity of compound 2 in vivo.
References:
1.Khan, M. I. et al. Send Orders for Reprints to reprints@benthamscience.ae The Therapeutic Aspects of the Endocannabinoid System (ECS) for Cancer and their Development: From Nature to Laboratory. Current Pharmaceutical Design vol. 22 (2016).
2. McDougle, D. R., Kambalyal, A., Meling, D. D. & Das, A. Endocannabinoids anandamide and 2-arachidonoylglycerol are substrates for human CYP2J2 epoxygenase. Journal of Pharmacology and Experimental Therapeutics 351, 616–627 (2014).
3. Bedse, G. et al. Therapeutic endocannabinoid augmentation for mood and anxiety disorders: Comparative profiling of FAAH, MAGL and dual inhibitors. Transl Psychiatry 8, (2018).
4. He, Y. et al. Discovery, synthesis and evaluation of novel reversible monoacylglycerol lipase radioligands bearing a morpholine-3-one scaffold. Nucl Med Biol 108–109, 24–32 (2022).