System Used:
M-Series

The global population is undergoing a significant demographic shift, with a rapid increase in the number of elderly individuals. This demographic trend poses new challenges in various aspects, including healthcare, social welfare, and economics. Age is a major risk factor for developing cardiovascular diseases, and heart failure (HF) is one of the leading causes of mortality worldwide. HF is a complex syndrome characterized by the heart’s inability to pump sufficient blood to meet the body’s demands.
While the relationship between aging and HF is well-established, the precise mechanisms linking these two processes remain unclear. Aging is associated with various structural and functional changes in the cardiovascular system, including alterations in cardiac function, increased fibrosis, impaired vascular function, and changes in gene expression. These age-related changes may contribute to the development and progression of HF.
Understanding the intersection between aging and HF is of paramount importance for developing effective therapeutic strategies to manage this debilitating condition. In this paper Berkowicz et al. aim to identify the functional, morphological, and transcriptomic changes associated with cardiac aging in a murine model of slowly progressing HF. This group, from Poland and Hungary, use a variety of imaging, and non-imaging approaches to investigate coronary microvascular dysfunction (CMD). Specifically, they used a unique murine model, Tgαq*44 mice, which exhibit a prolonged progression of HF that occurs at around 12-14 months of age. These mice were compared with wild-type FVB mice as a control group. The researchers employed a multidimensional approach, including functional assessments, morphological analysis, and transcriptomic profiling, to characterize the cardiac aging phenotype and its relationship to HF progression.
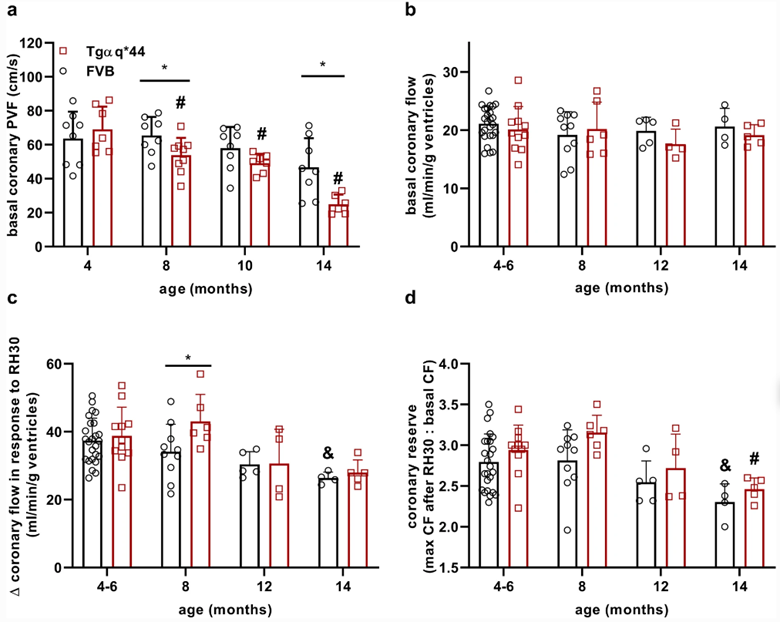
In the wild-type FVB mice, the aging process resulted in impaired diastolic cardiac function and reduced basal coronary flow (CF). These changes were accompanied by perivascular and interstitial fibrosis. However, there were no significant alterations in the cardiac activity of angiotensin-converting enzyme (ACE) or aldosterone plasma concentration, suggesting that other mechanisms contribute to the cardiac aging process in this model.
In contrast, the Tgαq*44 mice exhibited a progressive impairment of both systolic and diastolic cardiac function as HF advanced. These mice also displayed reduced basal CF, distinct rearrangement of the capillary architecture, pronounced perivascular and interstitial fibrosis, and activation of cardiac ACE and systemic angiotensin-aldosterone-dependent pathways.
Cardiac function was assessed using cardiac MRI, similar to what can be accomplished using the M-Series compact MRI system; while analysis was performed using the Segment software. Coronary flow (CF) was assessed in this study using the Doppler Flow Velocity System, which could also have been used to assess diastolic function in this study as well.
In addition to the functional characterization of this model, the researchers conducted a comprehensive transcriptomic analysis to identify age-related genes and processes that occur early in HF progression in Tgαq*44 mice. They identified 34 genes and eight biological processes associated with aging, which persisted throughout HF progression. These genes and processes were predominantly related to extracellular matrix remodeling and fibrosis, particularly perivascular fibrosis, leading to coronary microvascular dysfunction (CMD) in Tgαq*44 mice.
The findings of this study provide valuable insights into the role of accelerated cardiac aging in the pathophysiology of chronic HF. The Tgαq*44 mouse model allowed researchers to observe the manifestation of cardiac aging phenotypes during different stages of HF progression. The presence of robust perivascular fibrosis and extracellular matrix remodeling in the coronary microvasculature of Tgαq*44 mice indicates an accelerated cardiac aging phenotype that contributes to CMD and ultimately to the pathophysiology of chronic HF.
The study highlights the importance of CMD in the context of HF, particularly in HF with preserved ejection fraction (HFpEF), which is commonly observed in the elderly. CMD involves structural and functional alterations in the coronary microcirculation, including luminal narrowing, perivascular fibrosis, and impaired vasodilation. These changes can lead to myocardial ischemia and have prognostic implications in HF and other cardiovascular conditions.
Moreover, the study demonstrates that age-related genes and processes are not limited to the late stages of HF but are also evident early in the disease progression. The persistent activation of extracellular matrix remodeling and fibrosis-related pathways suggests their contribution to HF pathophysiology, emphasizing the need for targeted treatments that address these mechanisms.
In conclusion, the research findings support the notion that accelerated and persistent cardiac aging contributes to the pathophysiology of chronic HF. The study highlights the presence of prominent perivascular fibrosis in the coronary microcirculation, resulting in CMD, as an accelerated cardiac aging phenotype requiring specific therapeutic interventions in chronic HF. Understanding the complex interplay between cardiac aging and HF pathophysiology is crucial for developing effective strategies to manage and treat HF, particularly in the context of CMD. Further research is needed to elucidate the underlying mechanisms and explore potential therapeutic targets to improve outcomes in patients with HF.
https://doi.org/10.1007/s11357-022-00716-y
References:
- Berkowicz, P., Totoń-Żurańska, J., Kwiatkowski, G. et al. Accelerated ageing and coronary microvascular dysfunction in chronic heart failure in Tgαq*44 mice. GeroScience (2023). https://doi.org/10.1007/s11357-022-00716-y